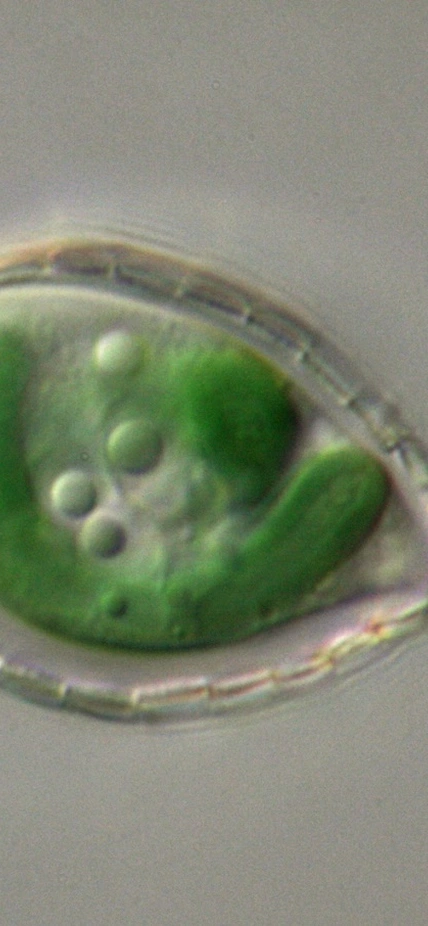
Palo Alto, CA— About 1.2 billion years ago a blue-green bacterium was engulfed by a more complex cell, transforming our planet and allowing a tremendous diversity of plant life to emerge and continue to evolve.
The engulfed cyanobacterium—sometimes called blue-green algae, because of its characteristic pigments —was capable of performing a process called photosynthesis, by which the Sun’s energy can be converted into chemical energy. At first, its relationship with the more-complex cell was symbiotic. It supplied the food and the other cell provided protection. Over time, however, much of the photosynthetic bacterium’s genetic material was transferred, little by little, to the more-complex “host” cell, until it was incapable of surviving on its own.
“This is the origin story for the generation of the cellular organelle responsible for photosynthetic activity, called the chloroplast,” explained Carnegie’s Victoria Calatrava. “A similar process resulted in the evolution of mitochondria, the so-called powerhouse of the cell, where energy is generated by breaking down carbohydrates and sugars.”
Although it is widely agreed upon to have occurred, there is still a great deal that scientists don’t understand about the process by which the genes were transferred, turning a symbiont into an organelle.
Calatrava, along with Carnegie’s Arthur Grossman and Devaki Bhaya, and in collaboration with Debashish Bhattacharya’s lab at Rutgers University, investigated this process by studying an organism called Paulinella. Their work was published by Proceedings of the National Academy of Sciences. This amoeba harbors a still-evolving organelle called a chromatophore, which was derived from a much more recent engulfment of a cyanobacterium that occurred only about 100 million years ago.
“Paulinella’s entire way of eating changed as a result of this event and now its nutrition is supplied by photosynthetic sugars made in the chromatophore,” Grossman explained. “As a result, this under-construction organelle is a great analog for understanding how a little cyanobacterium can evolve into a chloroplast—a game-changing event for our planet.”
The researchers showed that domestication of the cyanobacterium that is in the process of evolving into the chromatophore—and serving for research purposes as a stand-in for an ancient cyanobacterium’s evolution into the chloroplast—was facilitated by what scientists call a “copy-paste” mechanism named retrotransposition, which used RNA to make copies of the symbiont’s genes within the amoeba’s DNA and insert it at different positions in its genome.
The researchers found that the multi-site insertion resulting from the retrotransposition process benefitted the host amoeba by enabling it to take control of the cyanobacterial genes and increasing its ability to regulate its response to the stresses of excess light—when the cell would be highly susceptible to damage.
“The amoeba is a complex cell within the eukaryotic kingdom of the tree of life, and the symbiotic bacterium is a more primitive cell within the prokaryotic kingdom of the tree of life; understanding how the genes of the latter remained functional after being transferred between these two kingdoms of life is still an open question,” Bhaya noted.
Because it is an ancient mechanism that is highly conserved between organisms, it’s reasonable to believe that retrotransposition was critical for the evolution of the chloroplast and, by extension, it would underpin the existence of all plant life on Earth.
Acknowledgments
This work was supported by a grant from the National Aeronautics and Space Administration, the Carnegie Institution for Science, and a NIFA-USDA Hatch grant.