Baltimore, MD—Scientists have known for decades that certain genes (called transposons) can jump around the genome in an individual cell. This activity can be dangerous, however, especially when it arises in cells that produce eggs and sperm. Such changes can threaten the offspring and the success of a species. To ensure the integrity of these cells, nature developed a mechanism to quash this genetic scrambling, but how it works has remained a mystery. Now a team of scientists, including researchers at the Carnegie Institution’s Department of Embryology, has identified a key protein that suppresses jumping genes in mouse sperm and found that the protein is vital to sperm formation.
“There is a tiny cell component that is unique to germ cells—the precursors to egg and sperm—called nuage, which means ‘cloud’ in French. Other researchers recently suspected that nuage was involved in keeping genes from jumping around in germ cells of the female fruit fly,” explained Carnegie’s Alex Bortvin, a senior author of the study. “But until this mouse study, no one knew for sure if it was involved in mammalian germ cells. To test if the mouse nuage played a similar role in mammals, we focused on a mouse protein called Maelstrom whose distant relative protein in the fruit fly was implicated in the other study.”
In this research, published in the August 12th issue of Developmental Cell, the scientists first looked at where the protein Maelstrom resides during the formation of sperm. By marking the protein with a fluorescent antibody, they found that it was predominantly located in the cytoplasm, near the nucleus of the germ cell, at the nuage. To understand what Maelstrom does during the formation of sperm, the scientists created mutant mice that did not have the gene to produce the Maelstrom protein.
“We found that without the gene the process of meiosis was severely impaired,” said Bortvin. “There was a profound defect in interactions of parental chromosomes, a process known as synapsis, leading to death of germ cells. This was clear evidence that the protein is vital to the formation of sperm.”
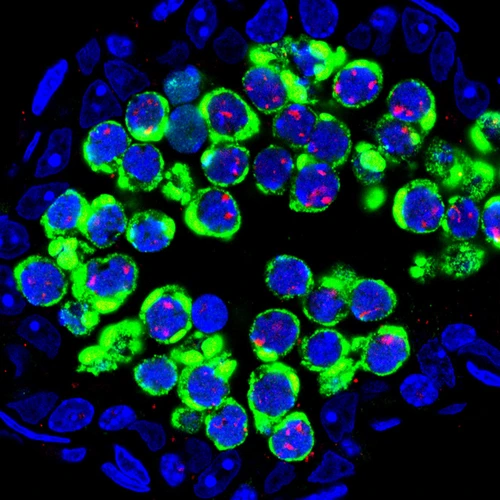
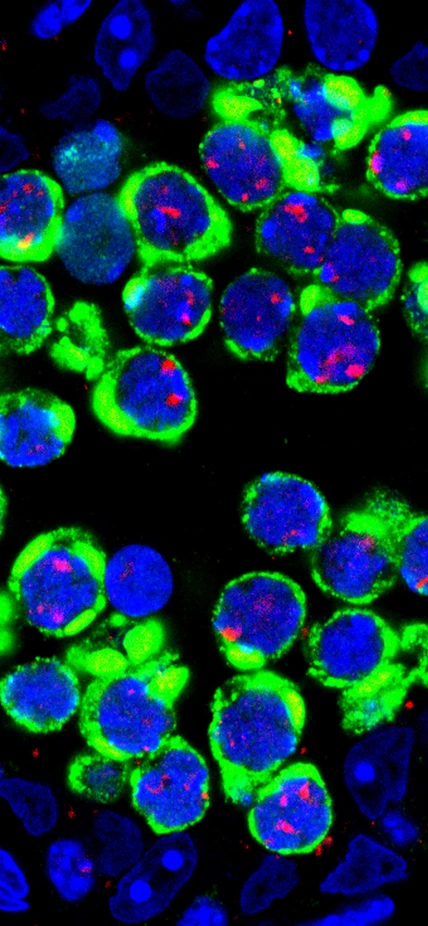
In the absence of the protein Maelstrom to keep them at bay, jumping genes, called transposons (green), flood germ cells (DNA-blue)—the precursor cells of sperm in the male mouse.
The cause of such a defect became apparent when the researches looked at the behavior of transposons. “We observed massive flooding of the cytoplasm and nuclei of germ cells by transposons in the mutant mice,” said Godfried van der Heijden, a Carnegie postdoctoral fellow and co-author. “This was the first time such a phenomenon was observed in germ cells of any species. Moreover, we found that the more transposons present in the nucleus, the more likely parental chromosomes would fail to locate each other during synapsis. Clearly, uncontrolled activity of jumping genes causes chromosomal mayhem in germ cells. Our results, coupled with work by Toshie Kai, a former Carnegie researcher studying the role of nuage in egg development in the fruit fly, suggest that nuage plays a central role in transposon silencing during the development of egg and sperm of many species from insects to mammals. ”
The last surprise for the scientists was the observation that, contrary to the current view in the field, the silencing of jumping genes does not occur one time only in male germ cells during the mouse fetal development. Instead, every time a germline stem cell divides by meiosis to make sperm in adults the jumping genes are activated only to be silenced soon thereafter.
“This was a very puzzling finding,” commented Bortvin. “Since the jumping genes are not silenced just once during the development of the fetus, but every time new sperm are produced during a mouse life, it’s possible that germ cells may employ transposons in some fundamental way in male germline meiosis. This research is the first such clue of that possibility. We will be very busy over the next few years trying to crack this and other puzzles of Maelstrom’s role in controlling meiosis and sperm production.”
See the video podcast at YouTube
This work was supported by the National Institutes of Health and the Carnegie Institution. Other authors of the study are Sarah Soper at The Johns Hopkins University; Tara Hardiman at Carnegie, Mary Goodheart with the Howard Hughes Medical Institute, the Whitehead Institute, and MIT; Sandra Martin at the University of Colorado School of Medicine; and Peter de Boer at Radboud University Nijmegen Medical Center, The Netherlands.